Study Consent Form Frequently Asked Questions
Blood Sample Collection Instructions
The SPRING Study: Severe acute resPiratory syndrome-Related coronavirus 2 prevalence In children and youNG adults in British Columbia: an observational study

Researchers at the Vaccine Evaluation Center would like to find out how many children and young adults in BC may have been infected with SARS-CoV-2 (the virus that was identified in December 2019 which causes the infectious disease known as COVID-19). The World Health Organization declared COVID-19 as a pandemic due to the rapid increase in the number of cases globally. Many cases in children, as well as in adults, have been mildly symptomatic or asymptomatic (without symptoms). This study will help researchers better understand the rates of COVID-19 infection amongst children and youth in BC and how they change as the pandemic progresses. As more people receive their COVID-19 vaccine, the study will also examine and compare antibody levels in vaccinated and unvaccinated individuals. This valuable information can help to guide policies and recommendations in BC for both the work and school environment.
Study Information
Who can participate?
Children and young adults under 25 years of age living in BC.
We are currently following up with existing SPRING participants to recruit for an extension of the SPRING Study. Recruitment for the extension is closed to the public.
What does the study involve?
Participants will be asked to answer a short online survey about their basic demographics and health, any symptoms of COVID-19, COVID-19 vaccination history, and their opinions on vaccines. They will also be sent a kit in the mail to collect an at-home self-administered finger/heel-prick blood sample.
How much of a time commitment is this study?
Less than 1 hour.
How will I receive the survey?
You will receive a unique survey link via email.
Why do you need a blood sample?
We will be able to test for the presence and amount of antibodies against the virus that causes COVID-19 using a small sample of your blood.
What is a dried blood spot (DBS) test?
A DBS test is a type of biological sampling that involves using a lancet to do a finger prick (or a heel prick if for infants under 12 months old), then blotting the blood onto a filter paper attached to a DBS card.

How will I receive and return the kit?
The kit will be sent to you by mail. To return the kit to our research laboratory, you can pack the completely dried DBS card in the postage-paid return envelope that we provide, and drop it in the mail.
Will I receive the results of the test?
You will be notified of your blood test results, which will show whether you have developed antibodies against the virus that causes COVID-19.
The tests being used in this study have been validated for research use only. While we will inform you if the tests are positive or negative, these are not a replacement for medical diagnostic tests for COVID-19. If at any time during this study you are recommended by a healthcare provider to have a test for COVID-19 or if you need a COVID-19 test for any other reason, you should still go ahead and have that test at a standard testing clinic. As well, if your antibody test is positive for COVID-19, that does not necessarily mean you are ‘immune’ or protected against future infections. You should continue to follow all public health guidance.
How do I sign up for this study?
Recruitment is closed to the public. We are currently following up with existing SPRING participants to recruit for an extension of the SPRING study.
If you have any questions, please click here and fill out an inquiry form. You can also email springstudy.vec@bcchr.ca or call the Vaccine Evaluation Center at 604-875-2187.
Resources for Participants
Downloads

Download and print instructions for finger prick.
Download and print instructions for heel prick.
Download tips for Reducing Your Child’s Discomfort During Dry Blood Spot Collection.
Watch our Dried Blood Spot Collection Instructional Videos
Finger Prick Instructional Video
Heel Prick Instructional Video
Frequently Asked Questions:
- 1. Who will use the data collected during this study and what for?
-
This study is being led by Dr. Manish Sadarangani and is funded by the Michael Smith Foundation for Health Research. The study team will use the results of this study to help doctors better understand the spread of COVID-19. Results of the study may be published online, in peer-reviewed journal articles, and presented at national or international conferences. Your participation will always remain strictly confidential, and your name will never appear in any publication of the results.
- 2. How many people will take part in this study?
-
There will be up to 16,000 children and young adults (under 25 years of age) in BC enrolled in this study.
- 3. Does this study involve in-person visits?
-
No. All study procedures will happen in your home. You will not come to any study centre. All materials will be sent to you by mail and the return mailing envelope will be postage paid.
- 4. How long will the online survey take to complete?
-
The survey will take between 10-15 minutes to complete.
- 5. Will it cost me anything to participate in this study?
-
There is no cost for taking part in this study
- 6. Will I receive compensation for participating in this study?
-
You will not be paid for taking part in this study.
- 7. Who can I contact with additional questions that are not addressed here?
-
If you have any questions or would like further information about this study before or during participation, you can contact the study coordinator at springstudy.vec@bcchr.ca or by calling 604-875-2422.
- 8. Am I eligible to participate if I receive the COVID-19 vaccine?
-
Yes, you can still participate in the SPRING Study.
- 9. I am a parent who wants to enrol my child on the study. Why do you need to speak with my child on the phone about this study?
-
For children between 7-13 years old, they will need to provide assent in order to participate in the study. Our research staff will need to give them a call to explain the study details, then assess and confirm their interest in participating in the study. The call will take around 10 minutes.
We recognize that some minors are able to make decisions for themselves. Generally, if a minor between the ages of 14-18 years old has reached a level of intellectual and emotional maturity such that s/he is capable of understanding and appreciating the nature and consequences of a particular decision, together with its alternatives, they can be considered capable of consenting. In this case, our research staff would give them a call to review study details and obtain their consent to participate in the study.
- 10. I signed and submitted my electronic consent form. When will I receive my survey?
-
Please allow up to 2 weeks to receive your online survey via email. You can contact us at springstudy.vec@bcchr.ca if you still haven’t received your survey after that.
- 11. Why can’t I access my survey link?
-
Please check your device’s firewall setting and see if it is blocking access to the REDCap website rc.bcchr.ca/redcap/. If yes, try temporarily disabling the firewall in order to access and submit the survey.
If you still can’t access the survey, please contact us at springstudy.vec@bcchr.ca
- 12. When will I receive my blood sample kit?
-
You will only receive this after you have completed and submitted your online survey. Please allow 2-3 weeks to receive your kit after you submit your survey.
- 13. I have multiple children participating in the study, but I only received the baseline survey/blood sample kit for one of them?
-
Depending on when their electronic consent/assent forms were submitted, their baseline surveys may be sent out at different time points. Receiving the blood sample kit also depends on when the online survey was completed.
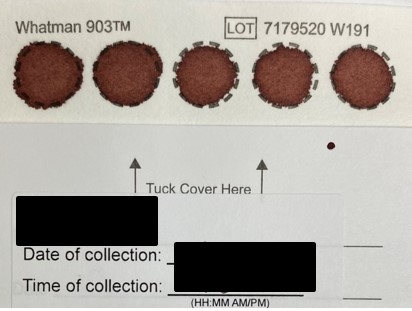
- 14. In the blood sample kit, there are two cards with circles on them. Which card should I be putting my blood sample on?
-
You should put your blood drops on the card that says “903 Protein Saver Card.” There is a folded cover on the card, has space for you to write your collection date and time, and has 5 circles for you to drop your blood onto.


Please do not drop your blood onto the humidity indicator card, which has 4 circles with blue/pinkish colour.

- 15. When will I receive the antibody results?
-
The research laboratory is currently testing the samples. It may take a few months to receive your antibody results. We appreciate your patience!
- 16. How will you be contacting me to inform me of my test results?
-
Your test results will be mailed to you when they become available.
- 17. I received multiple blood sample kits for my children. How do I differentiate between the dried blood spot cards?
-
The participant ID (PID) on the dried blood spot card should match with the number on the top right corner of the address label of the kit. The labels are also colour-coded to help you keep track of which dried blood spot card should be for which child.

If you are interested in joining one of our studies or
would like to be added to our contact list, click below.
